Here's our comparison of 7 manufacturing-relevant LIMS platforms (not ranked). We break down their strengths and trade-offs, and share cost and implementation expectations. Includes SampleManager, LabWare, STARLIMS, LabVantage, QBench, LabLynx, and 1LIMS.

Manufacturing labs don’t have the luxury of testing when there’s time. In many plants, a single delayed COA or missing result can slow down batch release, block shipping, and create costly production bottlenecks.
That’s why choosing a LIMS can make or break manufacturing operations.
1LIMS has conducted multiple LabCheck workshops with manufacturing laboratories, so we’ve decided to draw on our experience to create this guide.
We’ve compiled our experience into a list of the 7 best manufacturing LIMS, outlining where each solution performs well and where it falls short.
And yes, the LIMS we developed (1LIMS) is also included in the list. But we won’t describe it as the right choice in every case. Our goal is to help you make an informed decision, so you don’t waste time on a LIMS that doesn’t meet your needs.
Let’s move straight to our list of top 7 manufacturing LIMS solutions.
There is no such thing as the best-for-all LIMS for manufacturing. Every software comes with its own constraints. That’s why this list is not a ranking, but an objective selection of manufacturing LIMS solutions.
Further, we briefly introduce each LIMS to highlight where it performs well in manufacturing and where (inevitable) trade-offs exist.
1LIMS is built specifically for manufacturing QC labs that want to get rid of Excel, paper protocols, and outdated databases without turning LIMS implementation into a multi-year IT project. The platform is positioned for manufacturers (especially food & beverage) and service labs that need faster QC workflows, better traceability, and measurable ROI.
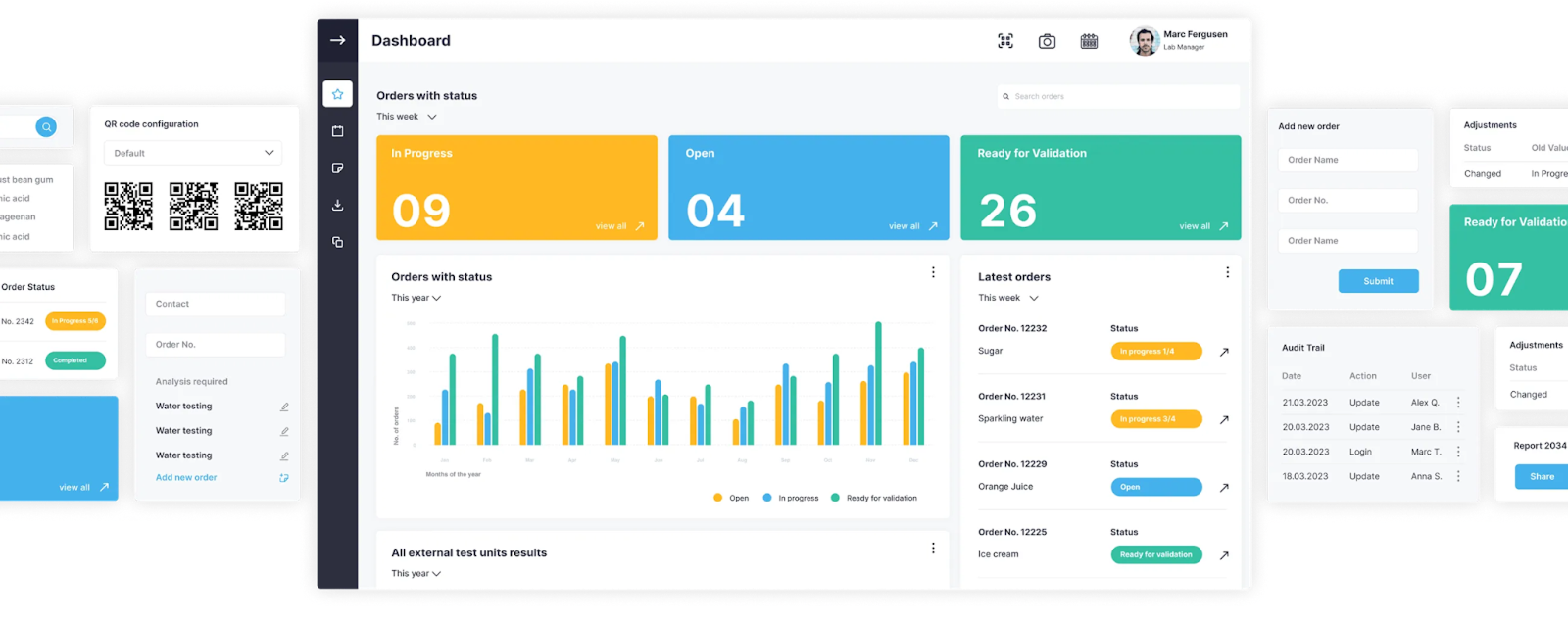
1LIMS works best for manufacturing labs that need to centralize all quality data and make QC processes faster and more consistent.
It focuses on manufacturing workflows such as:
A key part of the 1LIMS approach is the LabCheck workshop. Our team conducts a remote or on-site visit to map your current QC workflows, identify bottlenecks, and build a KPI-driven digitalization roadmap based on Lean Six Sigma methods (Gemba Walk, SIPOC, process quantification).
This is what we did for Model AG, a Swiss packaging manufacturer with plants across Europe. Their lab was still running on a mix of Microsoft Access, SAP, paper protocols, Excel files, and scanned PDFs.
During LabCheck, we analyzed their daily workload and found that lab staff spent 49 hours per week on quality data management, equivalent to 116% workload per 1 FTE.
By moving their QC workflows into a centralized LIMS, they could reduce workload by 50% and get estimated annual savings of up to CHF 41,067.
If labs have doubts on whether it’s rational to move to a LIMS, we can do a realistic calculation to see the exact result of LIMS implementation.
1LIMS isn’t suitable for companies that:
1LIMS is designed to complement ERP systems and connect QC workflows through integrations, but not to replace them.
To show where 1LIMS works best, here’s an example of a manufacturing company that reduced trend analysis from hours to 5 minutes.
Polygal AG is a Swiss manufacturer of natural hydrocolloids for food, pharma, and personal care that ships products to more than 55 countries worldwide. For years, Polygal managed this with a Microsoft Access database plus extra Excel sheets.
They came to 1LIMS to centralize all quality data and workflows in one system, and the results were impressive:
The results were so strong that Polygal expanded 1LIMS to sites in Spain, Pakistan, and China.
1LIMS has such cost ranges:
Implementation typically takes 1–3 months, with many labs operational in ~30 days.
1LIMS is a strong fit for manufacturing QC labs that want fast digitalization, reduced manual workload, and full traceability. It is best suited for teams that want measurable results quickly, rather than a long enterprise rollout.
SampleManager is one of the most common choices for an enterprise-grade LIMS. It’s built for regulated, high-throughput QC labs where the goal is controlling the full release process.
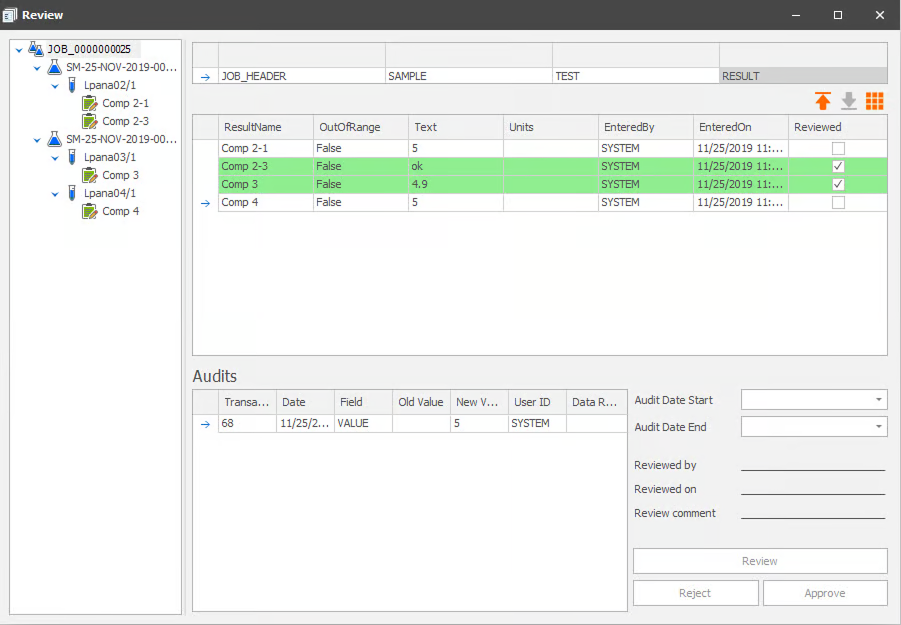
SampleManager isn’t a quick modern cloud tool. It’s a heavy-duty platform designed for manufacturers who need tight governance, traceability, and integration.
SampleManager works best for batch release and production decisions, not isolated testing. A major strength is its ability to connect QC workflows directly to ERP-driven processes, especially in SAP-based setups.
It also goes beyond a classic LIMS by bundling LIMS + SDMS + LES functionality, which is useful for companies that must prove SOP execution and compliance during audits.
The trade-off is the platform’s complexity. SampleManager is quote-based, usually requires an enterprise-level implementation approach, and can feel too heavy for smaller labs that just want to digitize basic QC reporting.

Full LIMS implementations often take 6–18 months, depending on integrations, migration scope, and validation requirements. Pricing is quote-based enterprise licensing.
SampleManager is a strong fit if your manufacturing lab runs on batch release, ERP workflows, and strict compliance. But if your main goal is quick deployment and flexibility, it may be too complex and costly than what you actually need.
LabWare is probably one of the most flexible enterprise-grade LIMS solutions. It’s a highly configurable platform that can be shaped to match complex QC and manufacturing workflows, if you’re willing to invest the time up front.
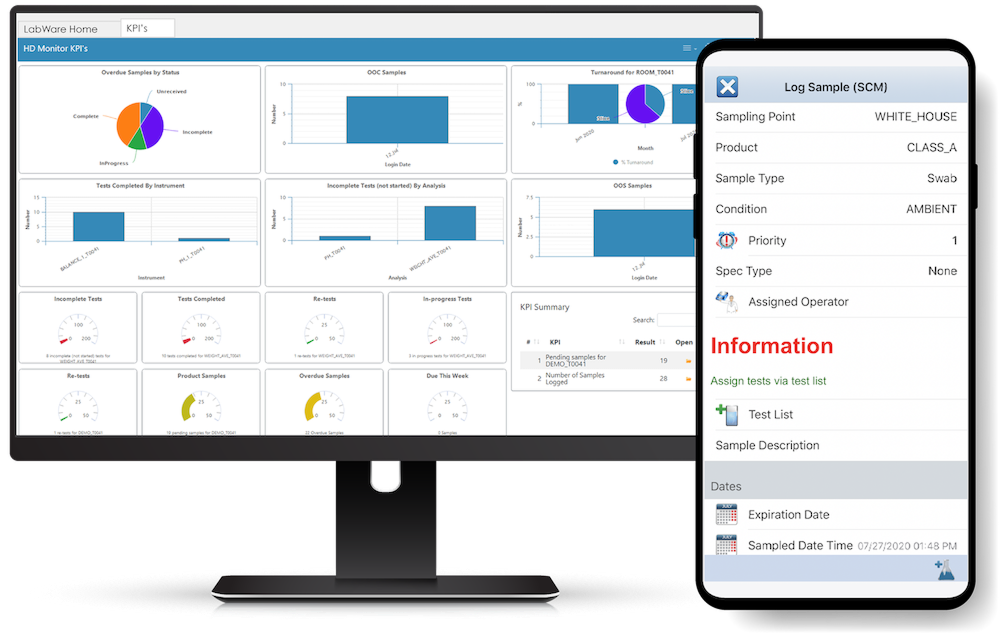
LabWare works best when you need deep workflow configurability and automation across diverse processes. It helps standardize complex quality tasks, supports traceability from sample receipt through results and reporting, and can be extended with modules like ELN and inventory management.
LabWare’s strength is its modular architecture and flexibility: you can tailor workflows without starting from scratch, which is especially valuable for manufacturers with varied QC processes.
The trade-off with flexibility is complexity and a learning curve. Many users note that setting up workflows and integrations (especially for instrument software) takes effort and expertise, while the system may feel overwhelming without support.
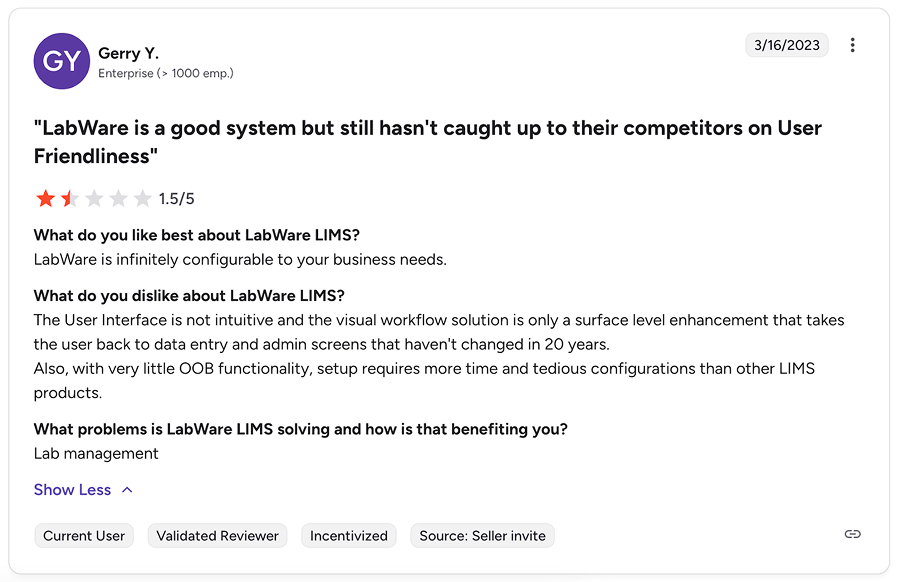
Some reviews also mention that deeply customized configurations can make future upgrades more challenging and that initial data modeling and project setup require solid planning.
LabWare does not publish standard pricing, and licensing is typically quote-based with variations by deployment model (on-premises vs cloud).
Implementation timelines for a fully configured LabWare LIMS vary from several months to over a year depending on workflow complexity, integrations, and validation needs.
LabWare LIMS is a strong choice if you need maximum configurability and automation and are prepared for an involved implementation. But for teams seeking fast deployment with minimal IT overhead, it can feel like more than what’s needed.
STARLIMS is a unified enterprise informatics platform. It’s designed not just as a traditional LIMS, but as a broader informatics suite combining LIMS with ELN, SDMS, advanced analytics, and mobile access.
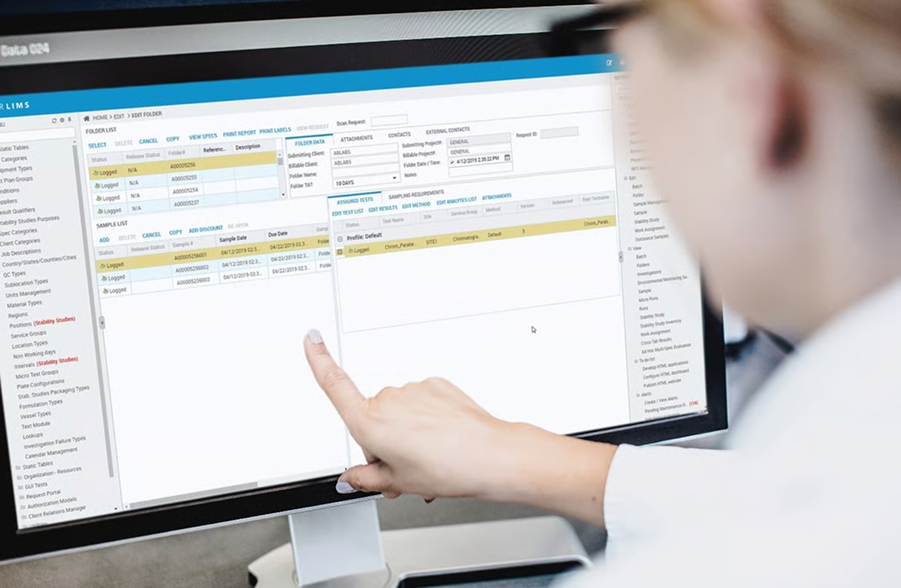
STARLIMS’s platform approach (integrating LIMS with ELN, SDMS, and analytics) helps break down data silos and gives visibility across the laboratory lifecycle.
Many users also praise its compliance-related functionality, including audit trails and electronic signatures.
The main trade-off is once again complexity. Because STARLIMS is a broad platform, implementation and configuration can take significant time and resources. Users note that it requires professional support to tailor workflows and get the most out of it.
STARLIMS does not publish standard pricing; its licensing is quote-based. Implementations can take several months.
STARLIMS is a solid choice for manufacturers that need strong traceability, compliance support, and centralized quality data across multiple sites. However, it is not the fastest system to implement, and it requires more setup effort than simpler LIMS tools.
LabVantage is one of the largest and longest-standing LIMS providers in the world, with a history dating back to the early 1980s.

LabVantage’s strengths are its broad capabilities and flexibility. The platform combines core LIMS features with ELN/LES/SDMS and analytics. LabVantage fits well for labs that want a single system to manage quality workflows, data capture, analytics, and compliance without assembling multiple point solutions.
LabVantage’s breadth is also its challenge. While it’s modular, the platform can become complex if your workflows are custom. Deep configuration and tailoring will require planning and, in many cases, professional support, especially with integration.

Some users also mention that initial setup and user training can take longer than with simpler LIMS, particularly if the project includes advanced analytics or intensive integration work.
Like most enterprise LIMS, LabVantage does not publish standard pricing; costs depend on deployment, modules chosen, and implementation scope.
Implementation time varies widely with complexity, a rollout can take multiple months.
LabVantage has broad capability and scalability, but companies should be ready for more configuration effort and learning curve compared with more narrowly focused LIMS options.
QBench is a cloud-based LIMS that aims to make laboratory management simple and flexible. It’s designed for labs that want to move quickly and avoid heavy custom coding.
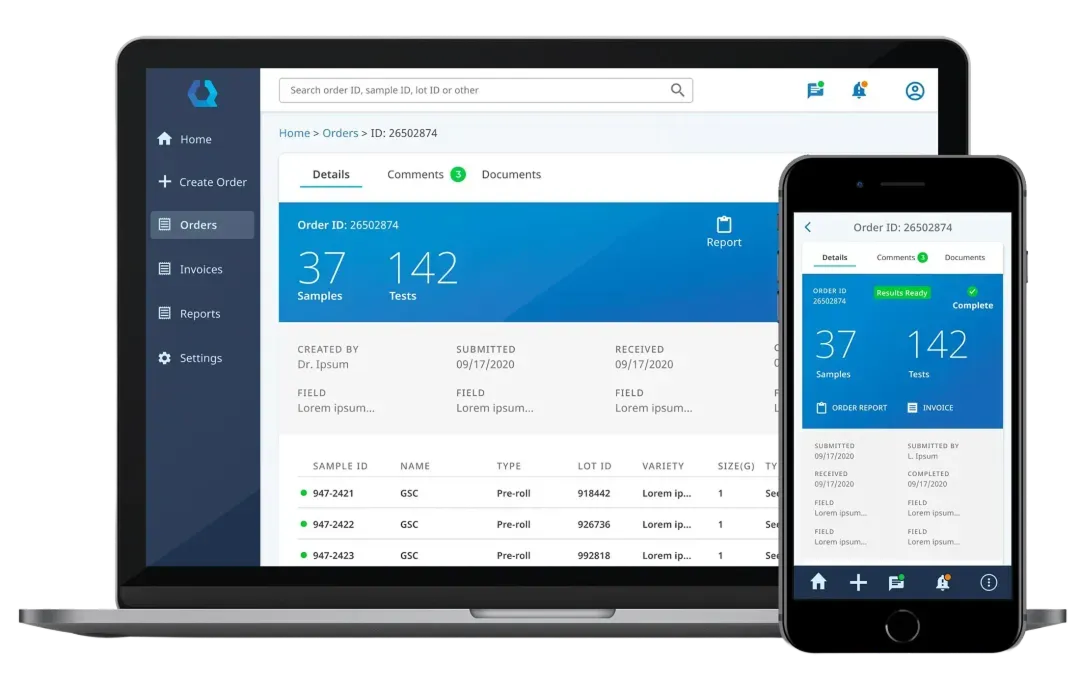
QBench stands out for being flexible and more user-friendly compared to many traditional LIMS solutions. It lets labs customize workflows, data fields, and reports without needing developers, which is a big plus for teams that want to adapt their system quickly.
QBench has limitations for complex manufacturing workflows:
QBench pricing is structured by annual plans, each including 5 users:
Enterprise: custom pricing

In terms of implementation, QBench is cloud-based and designed for no-code configuration, so rollout is typically faster than heavy enterprise LIMS platforms.
QBench can fit labs that want to replace spreadsheets and standardize basic QC workflows without a long implementation. It’s relatively easy to deploy, but companies with complex batch-release processes, heavy ERP/MES integration needs, or high-throughput production labs will run into limitations as workflows become more structured and demanding.
LabLynx was one of the first browser-based LIMS in the late 1990s, evolving into a flexible, configurable system used across many industries including manufacturing quality.
LabLynx is known for configurability and adaptability, letting labs tailor workflows, fields, and interfaces to fit unique needs rather than forcing a rigid template.
Because of its long history and modular design, LabLynx can be shaped to support a wide range of use cases, from batch QC workflows to clinical test management.
LabLynx is flexible, but that flexibility comes with trade-offs:
LabLynx does not publish fixed pricing; licensing is typically negotiated and can include a combination of subscription or perpetual models, user counts, modules selected, and hosting choices.
Implementation for configurable systems like LabLynx commonly takes 6–12 months.
LabLynx is a customizable LIMS platform with a long history, but implementation and configuration are more complex compared with newer systems.
There are dozens of LIMS platforms on the market, and the truth is simple: a system that works perfectly for one manufacturer can be a complete mismatch for another. Everything depends on your sample volume, batch release process, integrations (ERP/MES), compliance requirements, and how your lab operates day to day.
If you want an expert opinion before wasting months on research, book a short call with our team. We’ll quickly assess your situation and tell you honestly whether 1LIMS is a good fit, and if it isn’t, we’ll point you toward the type of solution that makes more sense.