Help shape the 2025 Lab Efficiency Report!
Take the survey now.
Your QC team is still on spreadsheets or using a LIMS nobody actually opens? This guide is for you. We cover why lab staff resist new software, what makes standard LIMS training ineffective, and what a faster, smoother adoption looks like in practice. You'll also see how Micarna Group went from paper chaos to processing 50% more samples in under four months, with zero training stress.

Switching to a Laboratory Information Management System (LIMS) is supposed to make analytical lab work easier. In practice, the first thing it makes harder is getting anyone to use it.
Lab staff resists new software because most LIMS are indeed hard to use. Forums and industry surveys tell the same story: implementations that dragged on for months, go-live dates that slipped, and staff who quietly went back to their spreadsheets.
There's a better alternative. At 1LIMS, we've helped manufacturing companies and service labs across Europe make the transition from spreadsheets and paper to a fully digital QC workflow in weeks. In this article, we'll share what drives adoption, what gets in the way, and the steps to take if you want to speed up LIMS implementation.
Ask anyone who's been through a LIMS implementation what the hardest part was, and they'll say getting their team to use it.
The complaints are everywhere. On Reddit, lab professionals describe implementations as nightmares, call specific systems difficult to learn, and even say they hate LIMS:
G2 and Software Advice reviewers flag the same issues: steep learning curves, workflows that feel worse than spreadsheets, and small tasks that somehow require navigating five screens. One reviewer noted that even printing a label required multiple screens. Another said that the LIMS they use is "NOT intuitive at all":

The numbers are even more telling. A director at a global biotech company described their LIMS as feature-rich but impossible to use in practice, even after abundant training. Adoption stalled at just 5% of available features. The rest of the team went back to paper.
And that's the core problem with how most LIMS vendors approach this. When adoption fails, the answer is always more training: more webinars, endless online sessions, a recorded walkthrough that nobody watches past the first ten minutes. But generic training doesn't fix a problem.
At 1LIMS, we've seen this pattern enough times to know that the problem lies in the approach. Here's what we do differently to make a LIMS transition fast and effective.
The way you solve the LIMS implementation problem determines whether your team embraces the system or quietly goes back to Excel. Let’s compare the standard approach and what you can do instead to help your lab adopt a new LIMS.
Most providers hand you a library of e-learning modules, schedule a few virtual sessions, and call it onboarding. The trainer has never seen your lab. The examples don't match your workflows. The slides were built for a standard out-of-the-box configuration, not the version you actually have.
The train-the-trainer model makes it worse. Vendors train a handful of "super-users," who are then supposed to transfer that knowledge to the rest of the team. In practice, those super-users become the only people who know the system, and the first ones everyone runs to when something goes wrong.
Before any configuration begins, 1LIMS runs a LabCheck workshop, an on-site process assessment using Lean Six Sigma methodology. The team maps your actual workflows, identifies bottlenecks, and defines the KPIs the implementation needs to achieve. Staff are involved from day one, not handed a finished system and told to adapt.
This is what happened at Estoppey-Reber SA, a Swiss producer of galvanic coatings. Their lab was running entirely on paper and Excel: 135 reports printed per week, with a technician spending over an hour each week just collecting data and generating reports.
Before recommending any software, 1LIMS conducted a LabCheck workshop directly at their facility, walking through every step of their process. After mapping the full picture, we defined what the software needed to do:
Now the company has a recommended feature set and knows exactly how much time and effort they could save with 1LIMS.
This matters for adoption more than any training session ever could. When people help shape the system, they don't resist it because it feels like their own.
Enterprise LIMS implementations routinely run three to twelve months. By the time the system is live, the team is exhausted, the initial enthusiasm is gone, and every small usability issue feels like a betrayal. Budget overruns are common. So is scope creep. Features that seemed necessary in month one become useless by month six.
1LIMS targets full onboarding in one month. The process is phased — assess, configure, test, go live — with deliverables at each stage. Configuration is matched to your actual workflows, not a generic template. And the commitment doesn't stop after you go live: post-implementation support is built in, so issues get resolved before they become habits.
Blerim Krasniqi, Member of the Executive Board at Hepart AG, put it plainly:
Most LIMS give every user the same dashboard, the same menus, and the same complexity, regardless of whether they're a lab technician scanning samples or a manager reviewing compliance reports. For the technician, that's a system full of features they'll never use and can't find the ones they need.
Role-based access in 1LIMS goes beyond security; first of all, it's a usability feature. Technicians see their workflows. Managers see their reports. Nobody has to scroll past irrelevant menus to do a routine task. That single design choice removes more friction than a full day of training.
The difference before and after 1LIMS implementation shows up in the numbers:
This is what happens when the implementation is built around the people who have to use it every day.
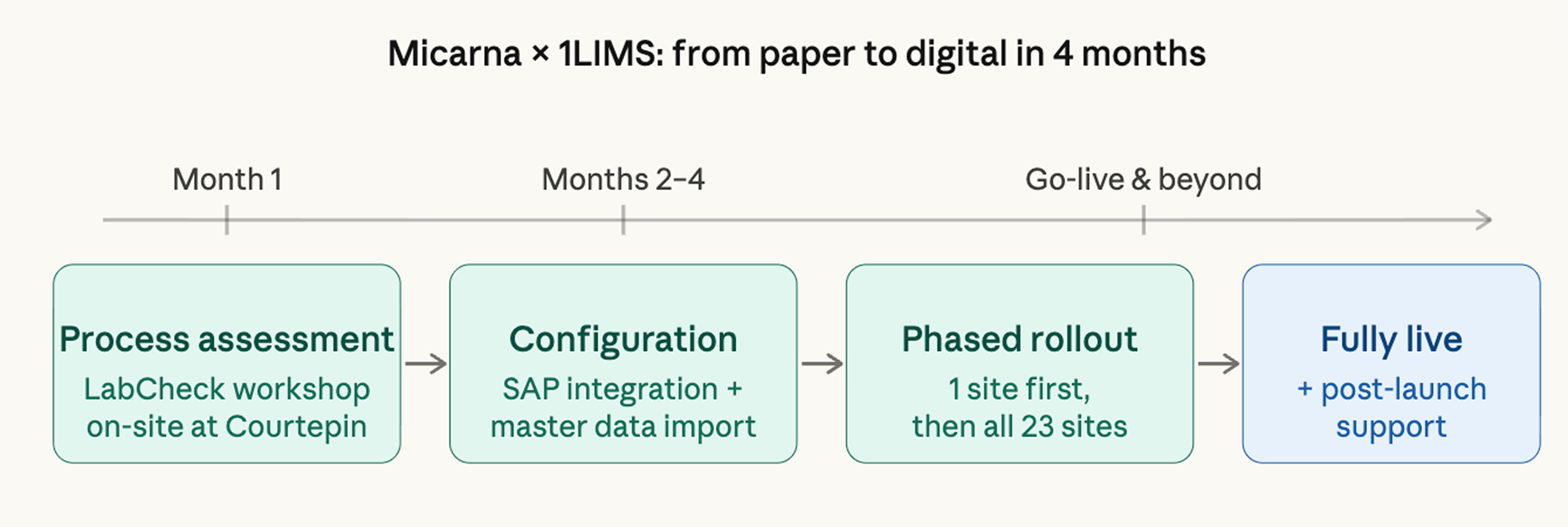
Finally, let’s see how 1LIMS implementation works in practice on the example of Micarna, a leading meat producer in Switzerland.
Micarna Group is Switzerland's largest meat, poultry, and seafood producer with 4,500+ products, 3,052 employees, and 23 production sites. Their QC lab was processing 100 to 200 samples daily with just three technicians. All of it was tracked on paper and Excel.
Stefania Cesarano, a Head lab manager, contacted 1LIMS to digitalize Micarna’s processes. That was the beginning.
Before a single screen was configured, the 1LIMS team sat down with Micarna's QC team to map how three technicians were managing 200 samples a day across 23 stakeholders. Master data, sampling plans, and SAP integration: all were defined upfront around Micarna's reality.
The result was a tailored solution instead of a standard package.
Rather than attempting a full rollout across all 23 locations at once, implementation started at the main production site in Courtepin, Fribourg. Issues were caught and resolved early, before they could replicate across the network. Only once the system was solid did the rollout expand, gradually, site by site.
Some of Stefania's technicians were barely comfortable with computers. It didn't matter.
"With just one or two clicks, you're exactly where you need to be. With other LIMS, I always found myself clicking through multiple screens to accomplish my goal," she said. The system worked the way the lab already thought.
For Stefania, the biggest daily win was eliminating the back-and-forth with 23 stakeholders sending in samples every day.
Before 1LIMS, compliance audits were, in Stefania's words, "a nightmare." Technicians under pressure would dig through spreadsheets looking for the right file, sometimes making errors in the process.
With 1LIMS, auditors accessed any batch result in two clicks. The audit trail was so clean and complete that accreditation assessors flagged the system as exemplary, which was a direct boost to Micarna's compliance standing.
Sample intake dropped from three to five hours each morning to two. Throughput increased by 50–60%. Data management became 50% faster. And with that reclaimed time, the lab was able to introduce advanced analytical techniques that hadn't been possible before.
None of this happened because Micarna's team sat through a webinar. It happened because the implementation was built around their lab from day one, and the system was intuitive enough that adoption took care of itself.
Most LIMS implementations fail because the vendor handed over a system, ran a few live QAs, and called it done.
Lab teams that have spent years with Excel and paper are resistant to being handed something complex and told to figure it out. That's a reasonable response. A two-hour online session built around a generic template has nothing to do with how their lab actually works. So they tolerate the training, nod along, and go back to what they know.
The fix isn't better webinars. It's a different approach entirely.
At 1LIMS, we don't send a login link and a help center URL. We come to your lab or schedule remote sessions. We walk the floor, map your workflows, talk to your technicians, and build the system around your lab’s workflows. If something isn't working post-go-live, we're still there to fix this.
You can start with a Labcheck workshop that will calculate the potential value of 1LIMS adoption, or go directly to 1LIMS implementation. In either case, you get a customized approach built for your workflows.